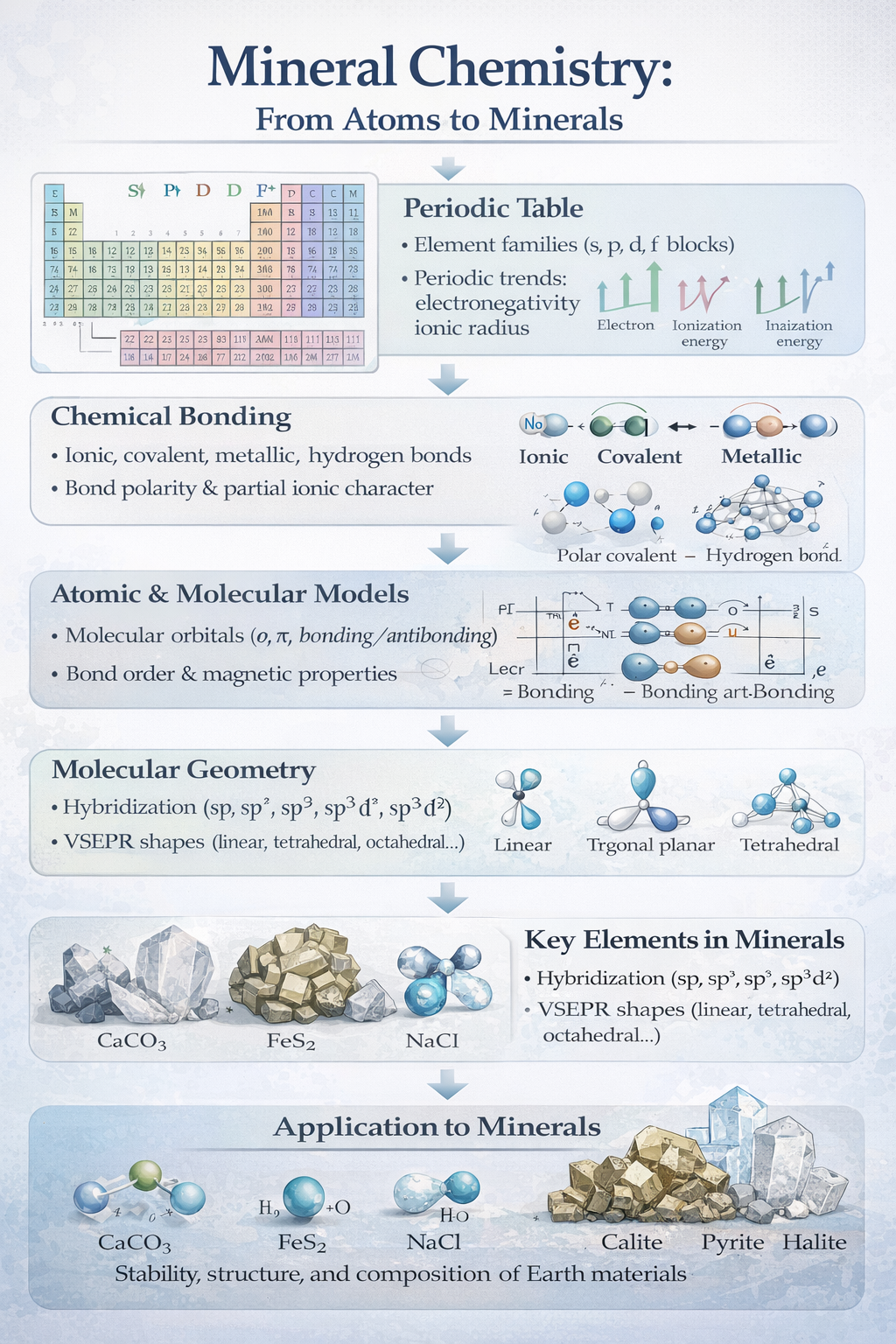
This second-year mineral chemistry course introduces the fundamental chemical principles governing minerals, with a strong focus on atomic structure and chemical bonding. It begins with the periodic table, emphasizing element families, periodic trends, and physicochemical properties such as ionic radius, electronegativity, and ionization energy. The course examines different types of chemical bonds—ionic, covalent, metallic, and weak interactions—and their role in mineral structures. Molecular orbital theory and bond energy concepts are used to explain mineral stability. Hybridization and VSEPR theory are introduced to understand molecular geometry. Special attention is given to major elements relevant to minerals, including hydrogen, oxygen, sulfur, halogens, and nitrogen. Their natural occurrence, chemical behavior, and common compounds are discussed. The course links atomic-scale properties to macroscopic mineral characteristics.
Examples and diagrams support conceptual understanding. Overall, the course provides a chemical foundation essential for advanced studies in mineralogy and geochemistry.
- معلم: BOURAS Hadj daoud
